  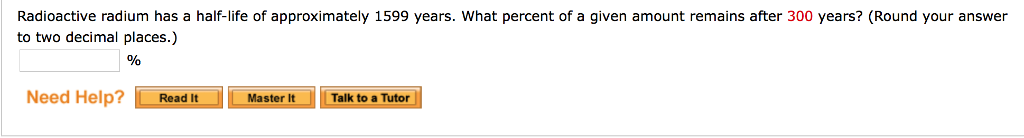
In this case, since 25 represents 1/4th of 100, two hal-life cycles must have passed in 1,000 years, since Let's say you started with 100 g and ended up with 25 g after 1,000 years. 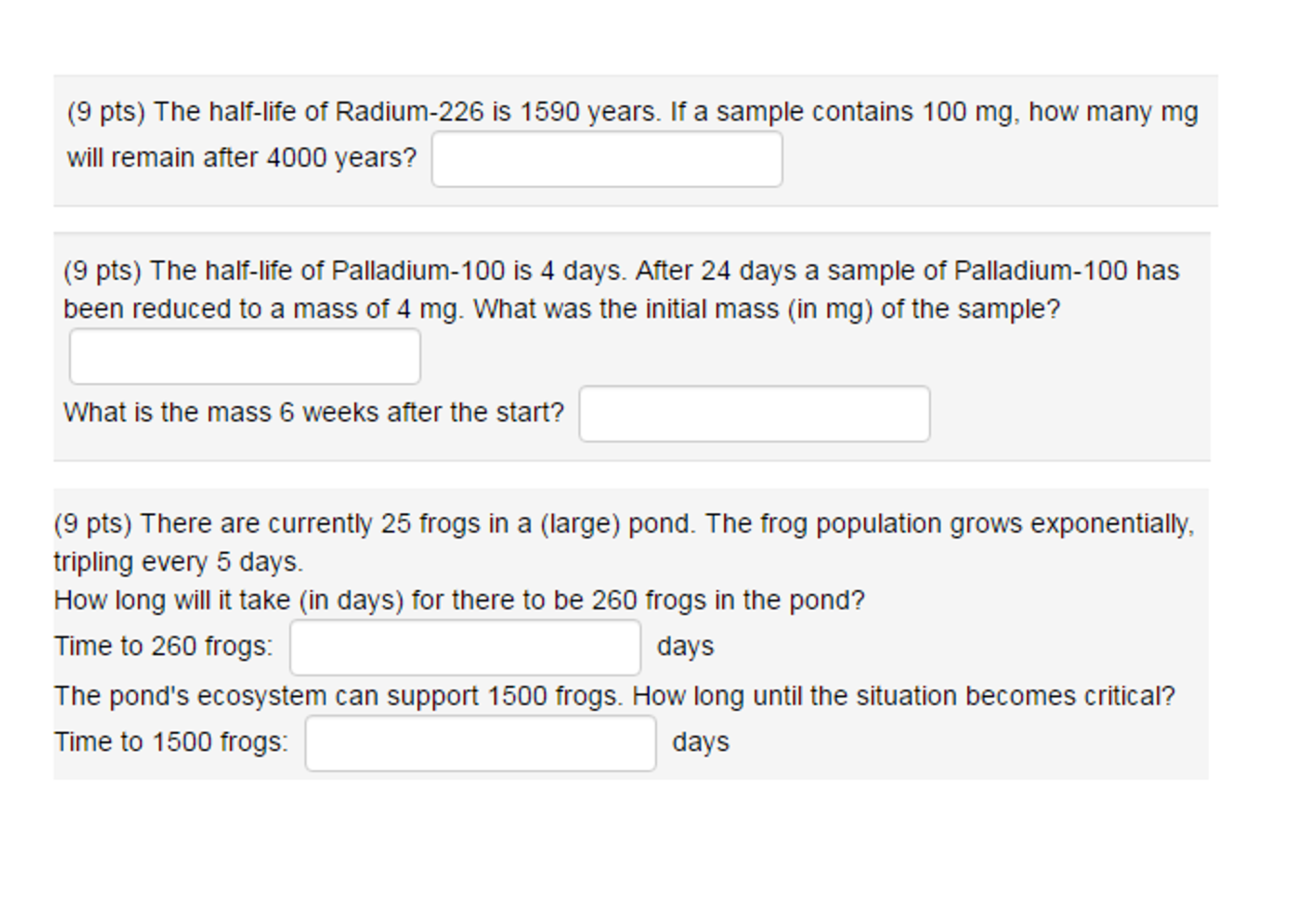
Sometimes, if the numbers allow it, you can work backwards to determine an element's half-life. So, the initial mass gets halved every 7.72 years. Therefore, its half-life is #t_("1/2") = 98.0/(12.7) = 7.72#"years"#. The amounts of radium-226 that decay during, and remain at the end of, each successive 1,600-year radium-226 half-life are symbolically-summarized below. Here's how you would determine its half-life: How many milligrams of a 15.0 mg sample of radium-226 remain after 6396 years if the half-life of radium-226 is 1599 years A radioactive element has a half-life of 5 years. It started from a mass of 67.0 g and it took 98 years for it to reach 0.01 g. half- life: as biological metaphor, 200, 213, 248, 317n21 origin of term, 19, 2327, 282n37, 291n32 probabilistic concept of, 12830, Hammer, William J. Let's say you have a radioactive isotope that undergoes radioactive decay. So, if a problem asks you to calculate an element's half-life, it must provide information about the initial mass, the quantity left after radioactive decay, and the time it took that sample to reach its post-decay value. If a sample contains 200 mg, how many mg will remain after 1000 years Problem 446RE: The half-life of radioactive iodine is 60 days. #t_("1/2")# - the half-life of the decaying quantity. The half-life of Radium-226 is 1590 years. half-life of 1620 years, and emitting alpha particles and gamma rays to form radon, and that is used chiefly in luminous materials and in the treatment of. #A_0# - the initial quantity of the substance that will undergo decay Exponential decay can be expressed mathematically like this: The fraction of radium atoms that remain undecayed after 4800 years will be. If half life period of radium is 1600 years, its average life period will be: View Solution. Nuclear half-life expresses the time required for half of a sample to undergo radioactive decay. The mean life of radium is: View Solution. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
